pH Levels and your Hydroponic System
pH Levels and your Hydroponic System
The two most important factors in water and nutrient management are pH and conductivity, and a good understanding and command of these variables is essential for successful hydroponic gardening.
The pH levels in hydroponic systems can often be overlooked if a grower is focusing more on monitoring a solution’s electrical conductivity or TDS level, balancing nutrients, providing beneficial additives and avoiding algae and plant pathogen problems.
Today, we’re going to be focusing primarily on pH.
pH is the measure of acidity or alkalinity of an aqueous solution. If a solution is acidic then it has a pH in the range of 0 to 6.9. If a solution is alkaline then it has a pH in the range of 7.1 to 14. Pure water or deionized water is neutral at pH 7.0. The ideal pH for most hydroponic systems application is between 5.8 and 6.2, except for Rockwool cultivation, which likes a slightly lower pH of about 5.5.
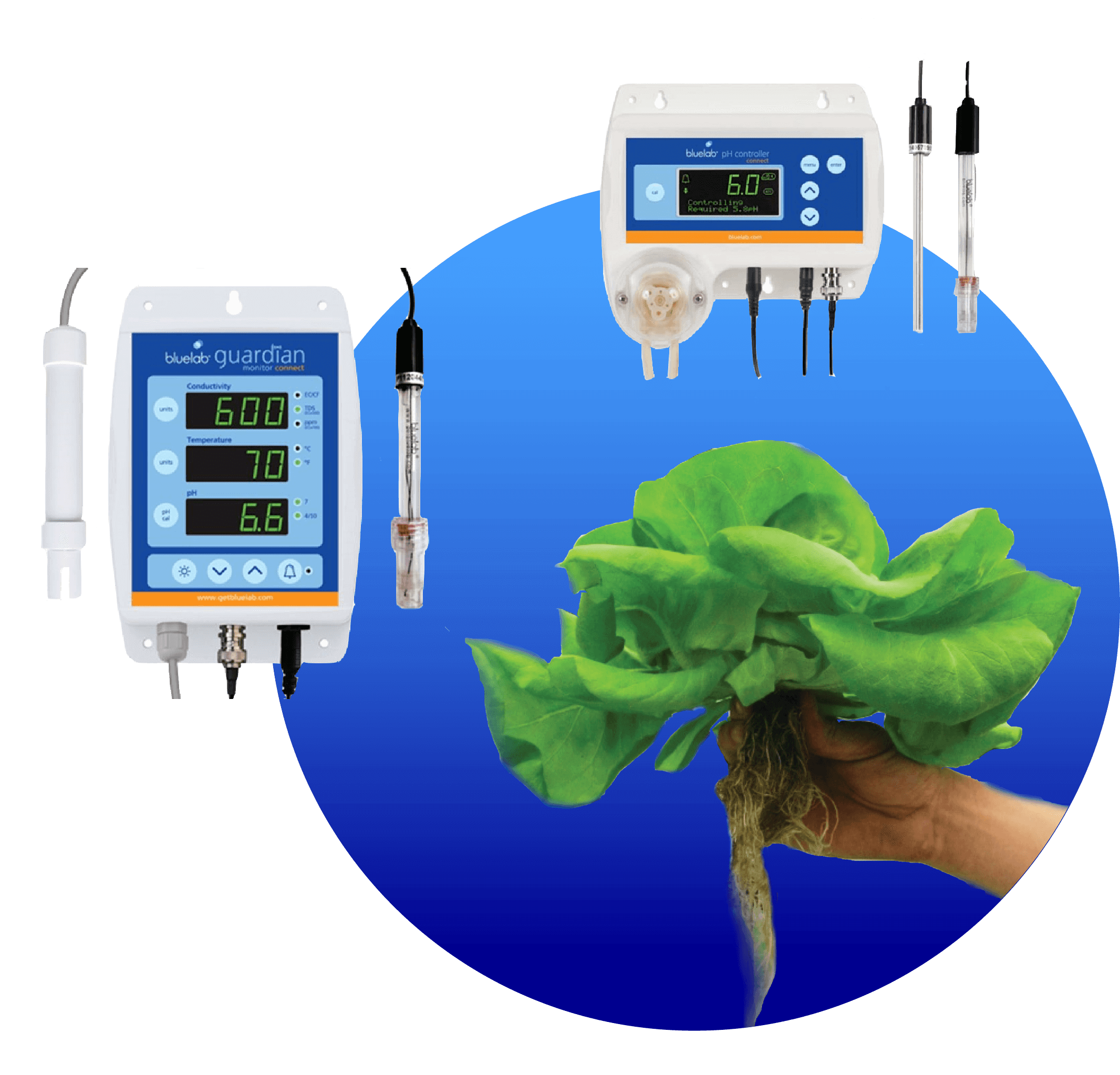
We are going to be focusing on the importance of pH in NFT Hydroponic systems. If the pH of a solution is not within the correct range the plant will not have the ability to absorb some of the essential elements required for proper plant growth. There are a few ways to check the pH of your nutrient solution, such as paper test strips, liquid pH test kits, and digital pH meters. We would highly recommend Bluelab if you were looking for digital pH instruments. We prioritize Hydroponic technology in India and the various Hydroponic kits that we sell in Chennai, where we’re based, and all over India have pH testing solutions included.
Automatic pH pens and meters are the most accurate for your hydroponic system, when calibrated correctly and offer continuous monitoring often with the possibility of data logging and taking into account temperature fluctuations, which can affect the calculation of pH levels.
Using a pH up and down the solution in small quantities allows for the control of the pH in your reservoir. Levels should always be checked after nutrient solutions have been added to the reservoir as they contribute to changes in the pH levels
